Adrenal glands play a critical role in maintaining hormonal balance through steroidogenesis, the process by which cholesterol is converted into steroid hormones. This energy-intensive process relies heavily on mitochondria, the cellular powerhouses responsible for generating ATP and facilitating key biochemical reactions. However, when mitochondrial function is disrupted—a phenomenon known as adrenal uncoupling—the consequences ripple through the entire endocrine system, leading to hormonal imbalances and systemic dysfunction. This article explores the mechanisms, causes, and implications of adrenal uncoupling, shedding light on the mitochondrial chaos at its core.
Adrenal Deal Breakers: Read More…
Mitochondria Require Oxygen and Glucose to Function
Mitochondria are the primary energy producers in the cell through cellular respiration, a process requiring both glucose and oxygen.
How it works:
- Glucose is broken down through a series of steps (including glycolysis and the citric acid cycle) to produce energy in the form of ATP (adenosine triphosphate).
- Oxygen acts as the final electron acceptor in the electron transport chain, a crucial step in ATP production.
- Without both glucose and oxygen, mitochondria cannot efficiently produce ATP, hindering the cell’s ability to perform vital functions.
Insulin resistance and disturbed delivery:
Insulin resistance affects the body’s ability to utilize glucose efficiently.
- Insulin promotes glucose uptake into cells, particularly in tissues like muscle, fat, and liver, so it can be used for energy or storage.
- In insulin resistance, cells don’t respond properly to insulin, leading to impaired glucose uptake and utilization.
- This can result in higher blood glucose levels (hyperglycemia), as the body struggles to get glucose into the cells that need it.
- While insulin’s direct effect on mitochondrial function is still being investigated, research suggests that insulin resistance can lead to mitochondrial dysfunction in various tissues, including muscle and liver.
- Mitochondrial dysfunction in insulin resistance may include reduced ATP production and increased production of reactive oxygen species (ROS), further impairing insulin signaling and potentially contributing to metabolic complications.
Therefore, insulin resistance can directly disrupt the “delivery” of glucose to the mitochondria by hindering glucose uptake into cells, thus impacting their energy production capabilities.
Recommended Test: 👉 BioScreen Sustain Max and move from guessing… to knowing.
The Role of Mitochondria in Adrenal Function
Mitochondria are central to adrenal steroidogenesis. The process begins with the conversion of cholesterol into pregnenolone, a precursor to all steroid hormones, including cortisol, aldosterone, and sex hormones. This initial step, catalyzed by the enzyme cytochrome P450scc (CYP11A1), occurs within the mitochondrial matrix and requires a tightly regulated supply of energy and reducing equivalents (NADPH). Mitochondria also maintain a proton gradient across their inner membrane, known as the mitochondrial membrane potential (ΔΨm), which drives ATP synthesis and supports the transport of cholesterol into the mitochondria via the steroidogenic acute regulatory (StAR) protein.
The efficiency of this process hinges on precise mitochondrial signaling and coordination with other cellular components. Any disruption to mitochondrial integrity or function can impair steroidogenesis, leading to reduced hormone production and downstream physiological consequences.
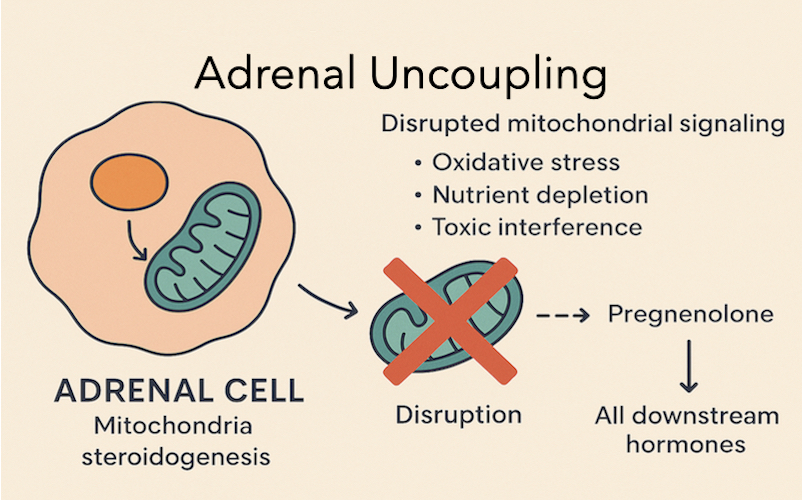
What is Adrenal Uncoupling?
Adrenal uncoupling refers to the disruption of mitochondrial signaling and function in adrenal cells, resulting in impaired steroidogenesis. Specifically, it involves the uncoupling of oxidative phosphorylation, where the proton gradient across the mitochondrial inner membrane is dissipated without generating ATP. This inefficiency reduces the energy available for cholesterol conversion and disrupts the delicate balance of reactive oxygen species (ROS) within the mitochondria.
When uncoupling occurs, the conversion of cholesterol to pregnenolone is hindered, creating a bottleneck in the steroidogenic pathway. As pregnenolone is the precursor to all adrenal hormones, this disruption cascades, leading to deficiencies in cortisol, aldosterone, and sex hormones, which can manifest as fatigue, immune dysfunction, metabolic disorders, and reproductive issues.
Sick and Tired of Being Sick and Tired?
Get the clarity your health deserves with the BioScreen Sustain Max blood test.
Whether you’re just beginning your wellness journey or following up on previous results, this advanced panel provides in-depth insight into your body’s most critical systems—far beyond standard lab tests.
What’s Included:
✅ Blood sugar & insulin function
✅ Cardiovascular and lipid health
✅ Hidden infections and inflammation
✅ Thyroid performance
✅ Anemia and electrolyte balance
✅ Liver and bile function
✅ Urinalysis with 13 key markers
Total: 61 biomarkers + 13 urinalysis evaluations—all in one comprehensive screen.
🔬 Take control of your health now.
Click below to order your BioScreen Sustain Max and start uncovering the hidden reasons behind your fatigue, inflammation, or stubborn symptoms.
👉 BioScreen Sustain Max and move from guessing… to knowing.
Causes of Adrenal Uncoupling
Adrenal uncoupling can be triggered by several factors, each contributing to mitochondrial dysfunction and chaos:
1. Oxidative Stress
Excessive ROS production, often caused by chronic stress, inflammation, or environmental toxins, overwhelms mitochondrial antioxidant defenses (e.g., superoxide dismutase and glutathione). ROS can damage mitochondrial DNA, proteins, and lipids, impairing the electron transport chain (ETC) and reducing ΔΨm. This oxidative damage disrupts the efficiency of steroidogenesis and promotes further ROS generation, creating a vicious cycle of mitochondrial dysfunction.
2. Nutrient Depletion
Steroidogenesis is a nutrient-intensive process requiring adequate supplies of cofactors such as coenzyme Q10, magnesium, and B vitamins (particularly B5, or pantothenic acid, a precursor to coenzyme A). Deficiencies in these nutrients, often due to poor diet or malabsorption, impair mitochondrial energy production and cholesterol transport. For example, insufficient coenzyme A limits the activation of fatty acids, which are critical for mitochondrial function in adrenal cells.
3. Toxic Interference
Environmental toxins, such as heavy metals (e.g., mercury, lead), pesticides, and endocrine-disrupting chemicals (e.g., bisphenol A), can directly target mitochondria. These toxins inhibit key enzymes in the ETC, disrupt membrane integrity, and interfere with cholesterol transport into mitochondria. For instance, mercury has been shown to bind to thiol groups in mitochondrial proteins, impairing their function and promoting uncoupling.
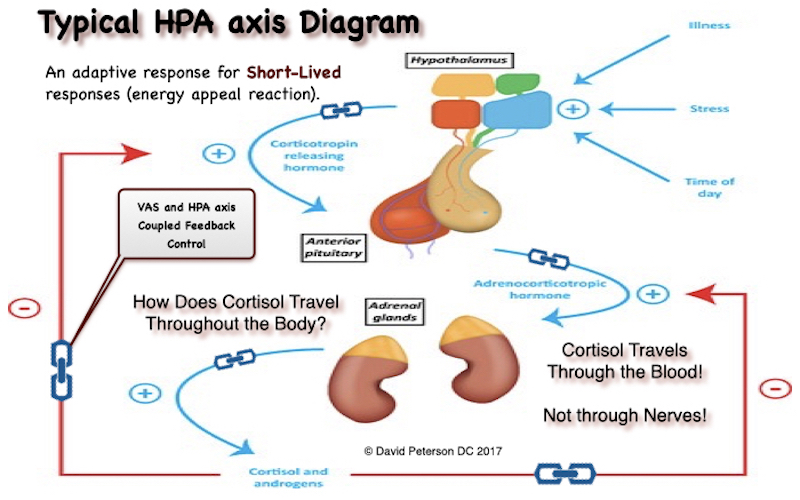
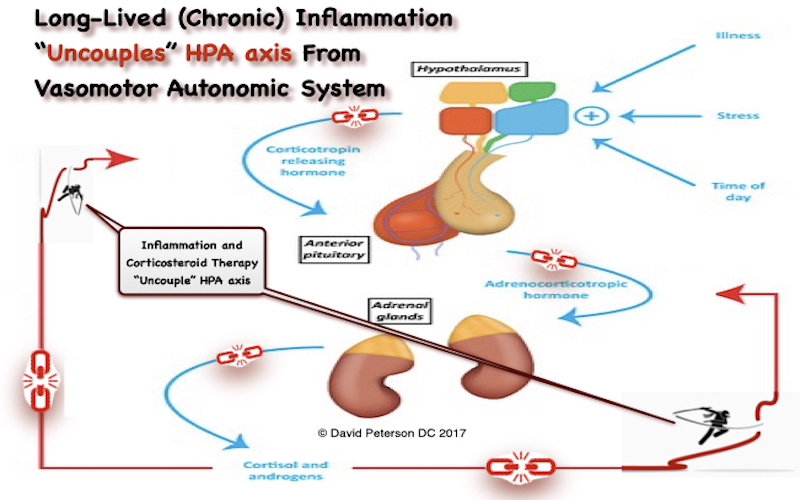
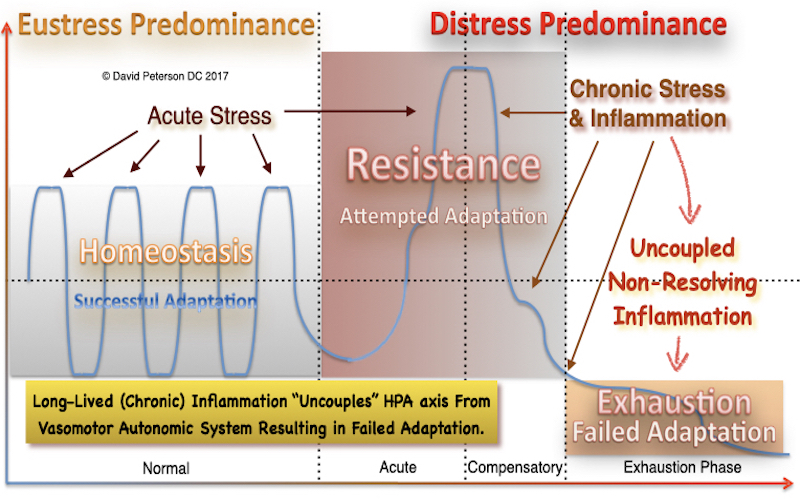
4. Chronic Stress
Prolonged activation of the hypothalamic-pituitary-adrenal (HPA) axis, as seen in chronic stress, places excessive demands on adrenal mitochondria. This overdrive increases ROS production and depletes mitochondrial resources, leading to uncoupling and eventual adrenal insufficiency. Chronic stress also upregulates uncoupling proteins (UCPs), such as UCP2, which dissipate the proton gradient and reduce ATP synthesis.
Consequences of Adrenal Uncoupling
The downstream effects of adrenal uncoupling are profound, affecting multiple physiological systems:
- Hormonal Imbalances: Reduced pregnenolone production limits the synthesis of cortisol, aldosterone, and sex hormones, leading to symptoms such as fatigue, salt cravings, hypotension, and reduced libido.
- Metabolic Dysfunction: Cortisol deficiency impairs glucose homeostasis, contributing to hypoglycemia or insulin resistance. Aldosterone deficiency disrupts sodium-potassium balance, leading to electrolyte imbalances.
- Immune Dysregulation: Cortisol is a key regulator of inflammation. Its deficiency can lead to unchecked inflammation or immune suppression, increasing susceptibility to infections and autoimmune conditions.
- Neurological Effects: Mitochondrial dysfunction in adrenal cells can exacerbate systemic oxidative stress, contributing to cognitive impairment, mood disorders, and neurodegenerative conditions.
Strategies to Mitigate Adrenal Uncoupling
Addressing adrenal uncoupling requires a multifaceted approach aimed at restoring mitochondrial function and reducing stressors. Potential strategies include:
- Antioxidant Support: Supplementation with antioxidants such as N-acetylcysteine (NAC), alpha-lipoic acid, and vitamin C can help neutralize ROS and protect mitochondrial components.
- Nutrient Repletion: Ensuring adequate intake of B vitamins, magnesium, and coenzyme Q10 supports mitochondrial energy production and steroidogenesis. Foods rich in these nutrients, such as leafy greens, nuts, and organ meats, should be prioritized.
- Toxin Avoidance and Detoxification: Minimizing exposure to environmental toxins and supporting detoxification pathways (e.g., through glutathione precursors or liver-supporting herbs like milk thistle) can reduce mitochondrial damage.
- Stress Management: Practices such as mindfulness, yoga, and adequate sleep can downregulate the HPA axis, reducing the burden on adrenal mitochondria.
- Mitochondrial-Targeted Therapies: Emerging therapies, such as mitochondrial-targeted antioxidants (e.g., MitoQ) or peptides (e.g., SS-31), show promise in protecting mitochondria from oxidative stress and restoring function.
Conclusion
Adrenal uncoupling represents a state of mitochondrial chaos with far-reaching implications for hormonal and systemic health. By disrupting the critical process of steroidogenesis, it creates a cascade of deficiencies that manifest as diverse symptoms, from fatigue to immune dysfunction. Understanding the causes—oxidative stress, nutrient depletion, toxic interference, and chronic stress—provides a framework for targeted interventions. By supporting mitochondrial function through nutrition, stress management, and toxin avoidance, it is possible to restore adrenal health and mitigate the chaos within. Future research into mitochondrial-targeted therapies may offer new hope for addressing adrenal uncoupling and its consequences.
Adrenal Rehabilitation Support Products